World’s Smallest Antenna Can Measure Protein Changes
World’s Smallest Antenna Can Measure Protein Changes
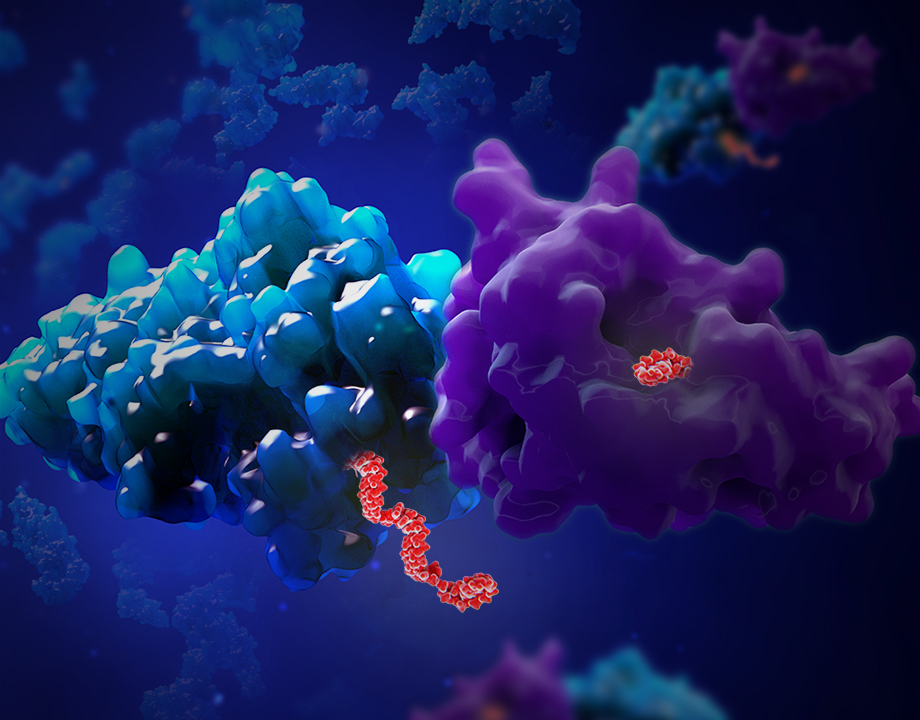

The antenna, made of DNA, will soon tell us big things about what goes on within complexly folded biomolecules.
At the root of many biological processes—and diseases—are changes in protein structures. Though our understanding of those structures has advanced leaps and bounds in the past decades, biomedical researchers have had no way of monitoring changes in real time.
That’s all about to change, thanks to a tiny antenna made of DNA produced by researchers out of the University of Montreal in Quebec.
The antenna, the world’s smallest, sprang from an earlier attempt to make a similarly tiny DNA thermometer.
“DNA unfolds with temperature,” said Alexis Vallée-Bélisle, a professor of chemistry at the university, and co-author of the paper “Monitoring Protein Conformational Changes Using Fluorescent Nanoantennas” which appeared in Nature Methods this January. “What could be the smallest system where we measure a change in temperature? We thought of enzymes.”
More Like This: DNA Data Storage
Vallée-Bélisle put fluorescent compounds—fluorophores—next to enzymes in the hopes of seeing a brighter glow when the enzymes loosened with heat. Indeed, the thermometer appeared to work: the fluorophores seemed to brighten with a change in temperature.
“Eighty percent of the people we showed it to were fascinated,” Vallée-Bélisle said. “But the physicists were saying, ‘I don’t think it’s possible—temperatures diffuse too fast.’ ” It turned out that the fluorophores were not reacting to temperature. “We were disappointed, but we realized that we could measure conformational change.”
Using the same method, Vallée-Bélisle and his colleagues created a system to bind fluorophores to different locales on any protein. Now, when there’s a change on a protein coupled with a fluorophore, the antenna will fluoresce within milliseconds.
“When the protein moves, it changes the structure of the fluorophore, and we are we are able to see the change,” he said. And that change can be detected with the kind of spectrometers found in labs everywhere.
The antennae could soon change the method, and speed, with which cancer drugs are developed. The mutation of some proteins, for example, can lead to certain cancers. To find a drug that binds to those proteins and returns them to their original state, researchers need a way to monitor the proteins. “That’s where a lot of time and money get lost,” Vallée-Bélisle said.
Bioengineering in Depth: Neat Little Packages
Current methods of developing such drugs look at huge libraries of possibilities—hundreds of thousands of drugs—hoping to find a match to what might be going on.
“If your car engine has a defect in it, if you just look at a picture, you aren’t going to detect it. You have to start it, and listen to it, and then an expert may say, ‘Oh, that’s your problem.’ ”
With repeated views of an enzyme’s activity, it will be possible to recognize a fluorescent signature. So, over time, and with continued use of the antennae, drug developers and microbiologists will have a greater understanding of exactly what is going on in proteins of interest, and when.
Selected for You: Building Tough 3D Nanomaterials
By using a 96-well plate, Vallée-Bélisle’s lab was able to create multiple antennae at one time, with different lengths, structures, and fluorophores. “Attaching the antennae next to the proteins is much simpler using this approach,” he said.
Labs that hoped to use the antennae could order plates of them and watch for fluorescence in their own labs with their own spectrometers.
“That will make the whole thing much much faster than if we have to select antennae for everyone in the world,” Vallée-Bélisle said. Once it’s been commercialized and has become standard practice, the tiniest antennae may make the biggest impact.
Michael Abrams is a science and technology writer based in Westfield, N.J.
That’s all about to change, thanks to a tiny antenna made of DNA produced by researchers out of the University of Montreal in Quebec.
The antenna, the world’s smallest, sprang from an earlier attempt to make a similarly tiny DNA thermometer.
“DNA unfolds with temperature,” said Alexis Vallée-Bélisle, a professor of chemistry at the university, and co-author of the paper “Monitoring Protein Conformational Changes Using Fluorescent Nanoantennas” which appeared in Nature Methods this January. “What could be the smallest system where we measure a change in temperature? We thought of enzymes.”
More Like This: DNA Data Storage
Vallée-Bélisle put fluorescent compounds—fluorophores—next to enzymes in the hopes of seeing a brighter glow when the enzymes loosened with heat. Indeed, the thermometer appeared to work: the fluorophores seemed to brighten with a change in temperature.
“Eighty percent of the people we showed it to were fascinated,” Vallée-Bélisle said. “But the physicists were saying, ‘I don’t think it’s possible—temperatures diffuse too fast.’ ” It turned out that the fluorophores were not reacting to temperature. “We were disappointed, but we realized that we could measure conformational change.”
Using the same method, Vallée-Bélisle and his colleagues created a system to bind fluorophores to different locales on any protein. Now, when there’s a change on a protein coupled with a fluorophore, the antenna will fluoresce within milliseconds.
“When the protein moves, it changes the structure of the fluorophore, and we are we are able to see the change,” he said. And that change can be detected with the kind of spectrometers found in labs everywhere.
The antennae could soon change the method, and speed, with which cancer drugs are developed. The mutation of some proteins, for example, can lead to certain cancers. To find a drug that binds to those proteins and returns them to their original state, researchers need a way to monitor the proteins. “That’s where a lot of time and money get lost,” Vallée-Bélisle said.
Bioengineering in Depth: Neat Little Packages
Current methods of developing such drugs look at huge libraries of possibilities—hundreds of thousands of drugs—hoping to find a match to what might be going on.
“If your car engine has a defect in it, if you just look at a picture, you aren’t going to detect it. You have to start it, and listen to it, and then an expert may say, ‘Oh, that’s your problem.’ ”
With repeated views of an enzyme’s activity, it will be possible to recognize a fluorescent signature. So, over time, and with continued use of the antennae, drug developers and microbiologists will have a greater understanding of exactly what is going on in proteins of interest, and when.
Selected for You: Building Tough 3D Nanomaterials
By using a 96-well plate, Vallée-Bélisle’s lab was able to create multiple antennae at one time, with different lengths, structures, and fluorophores. “Attaching the antennae next to the proteins is much simpler using this approach,” he said.
Labs that hoped to use the antennae could order plates of them and watch for fluorescence in their own labs with their own spectrometers.
“That will make the whole thing much much faster than if we have to select antennae for everyone in the world,” Vallée-Bélisle said. Once it’s been commercialized and has become standard practice, the tiniest antennae may make the biggest impact.
Michael Abrams is a science and technology writer based in Westfield, N.J.











