Is 3D Printing the Future of Manufacturing?
Is 3D Printing the Future of Manufacturing?


Many companies turned to additive manufacturing as COVID-19 disrupted global supply chains. Can additive manufacturing become the future of manufacturing? Photo: Research Institute CII RC CVUT
Clara Remacha, the strategic accounts project manager for HP’s 3D Printing and Digital Manufacturing business based in Barcelona, is an industrial engineer who works with hospitals and physicians to print 3D anatomical models and a variety of orthotics. She was part of the team that launched HP’s first 3D printer and has helped several customers turn 3D-printed designs into product applications over the last four years.
At a networking event at a local hospital in March 2020, a physician colleague of Remacha’s warned her about how COVID-19 was ramping up. More than 1,200 cases had been confirmed since the first case on January 31—barely over a month earlier. The doctor suggested that everyone should work from home and go remote. Sure enough, Spain enacted a lockdown the very next week.
What happened next in Spain became a reoccurring theme all over the world. Supply chains were disrupted, resulting in a lack of goods for consumers and raw materials for industry. Hospitals were flooded with COVID-19 cases, and doctors were forced to choose which patients would get a ventilator because of limited supply. Healthcare workers and the general public alike scrambled for personal protective equipment. It was a nightmare.
Remacha and her team soon started to receive requests from local hospitals to 3D print equipment. Juan Angel Muñoz López, a nursing supervisor at Príncipe de Asturias Hospital in Alcalá de Henares just outside of Madrid, reached out to Barcelona’s 3D Printing and Digital Manufacturing Center of Excellence. “He told us they had already reached the point where they had to decide who lived and who didn’t. They didn’t have enough respirators for everyone,” Remacha said. Muñoz wanted HP’s help to fabricate a part the ICU nursing staff had designed that could connect multiple standard elements to create a continuous positive airway pressure (CPAP) device. The CPAP device could help increase a patients’ blood oxygen levels above 90 percent, potentially sparing them from using a ventilator.
Within two days, Remacha and the HP team had the design evaluated, printed, and sent back to the hospital for validation. And within just two weeks, the CPAP cross-connector device was approved by the Agencia Española de Medicamentos y Productos Sanitarios, the Spanish medical device regulator.
Stories like these threw a spotlight on additive manufacturing and its potential for fast, flexible production of critically needed parts. Whether it was face shields, nasopharyngeal swabs, or respiratory masks, additive manufacturing rose to the challenge to fill in the gap caused by a disrupted supply chain. Now, some industry observers are starting to ask whether 3D printing could be the future of manufacturing.
Disrupted supply chains led several hospitals to ask additive manufacturing companies for help by producing potentially life-saving equipment. For companies like HP, it was a challenge on how to begin.
“We had no idea what was needed,” Remacha said. “We design 3D printers. We don’t design medical devices.”
HP’s first step was to set up an online form through which customers and users could submit requests for 3D-printed accessories to help them battle COVID-19 in their community. At first, people asked for hands-free door openers and mask adjusters. As the pandemic went on, healthcare workers started to ask for face shields. Remacha and her team had a virtual meeting with one of the doctors they work with to formalize the design on how to construct a face shield properly.
“We didn’t know what requirements we had to meet. The healthcare workers were now working 12-hour shifts, and they told us that comfort was one of the most important factors,” Remacha said.
The HP team met on a Saturday and by Monday had designed and printed a face shield prototype with an adjustable band. The team used taxis to ship parts back and forth between the hospital to ensure fit and functionality. By the following Friday, the design was on its fourth iteration. The design received full certification based on the European EN-166 standard a few weeks later.
Recommended for You: Lakshmi Vendra Reviews AM’s Use in Oil and Gas
Since announcing their efforts to help the medical community, HP and its global community partners produced more than 6 million parts for medical applications.
The ease of regulations and standards aided the additive manufacturing industry significantly during the pandemic. In the U.S., the Food and Drug Administration granted emergency use authorization (EUA) to 3D print parts for medical applications. For instance, Somerville, Mass.-based Formlabs was given an EUA to print and ship bi-level positive airway pressure (BiPAP) adapters to hospitals throughout the U.S. to combat ventilator shortages at the beginning of the pandemic.
America Makes is one of the nation’s leading and collaborative partners in additive manufacturing and 3D printing technology research and innovation. At the very start of the pandemic, America Makes, along with their partner network, documented a series of issues surrounding the supply chain and presented those to the commissioner of the FDA.
“Within days of the pandemic starting, we told the FDA that the AM community is ready to help. They’re sitting on the sidelines and can’t get engaged because they don’t know answers to these various [regulatory] questions,” said John Wilczynski, executive director at America Makes. “Almost immediately, the formation of an interagency agreement, between the Veterans Health Administration (VA) office, the FDA, and the National Institute of Health (NIH), all started to work together. There was a tremendous flow of information that was disseminated to the AM community, which helped manufacturers get started.”
The pandemic has encouraged hospitals to consider on-premise or on-demand manufacturing. Systems like the VA have taken significant strides in providing point of care device manufacturing, such as on-premise 3D printing. According to Wilczynski, the pandemic presents an opportunity to reconsider the supply chain and how parts are manufactured.
“In most cases, people haven’t gone down that path,” Wilczynski said. “The procurement system associated with healthcare does not provide the freedom to buy a single $50 item, which normally costs $1 for daily use, but with AM, we can. It’s an interesting idea because we are shifting from a consumable approach to a reusable approach.”
3D Systems in Rock Hill, S.C., partnered with the VA to design medical devices and take them through FDA clearance. The two organizations will use AM solutions to provide point of care, next-generation medical devices.
The initiative spurred out of the initial collaboration to address the lack of face masks within the VA hospitals at the start of the pandemic.
The next design was to develop a 3D-printed nasopharyngeal (NP) swab that could be manufactured on production-level equipment. The shortage of NP swabs was a significant hurdle as large-scale testing began to ramp up. 3D Systems were able to test and validate a custom medical-grade NP swab design out of DuraFrom ProxX PA using its ProX SLS 6100 printer. The material is a strong, tough thermoplastic material suitable for select medical devices such as NP swabs. The company was able to produce 3,000 swabs every 24 hours per printer.
“What began during the pandemic in response to a critical need has expanded to change the way healthcare is delivered,” said Menno Ellis, executive vice president of healthcare solutions at 3D Systems. “This is a one-of-a-kind collaboration between an additive manufacturing solutions provider and one of the world’s largest integrated health care systems to accelerate innovation in the medical device production and deployment arena.”
3D Systems has had plenty of success in delivering on-demand medical additive manufacturing applications within the dental industry. Dental appliances, including crowns, bridges, and dentures, are produced using additive manufacturing at a high volume and on-demand. For example, the dentist would take an intraoral scan and create a digital impression that is instantly analyzed. If a 3D printer is on-site, the dentist could manufacturer a try-in mold the very same day and test it on the patient. The patient takes the mold home to try for a few days before finalizing the dental fixture. The entire process cuts down what used to take weeks into a matter of days.
“We are seeing increasing use of patient-specific instruments and medical devices, and expect this will continue to accelerate,” Ellis said. “This is where 3D printing shines and differentiates itself from other technologies, because you have the benefit of producing in units of one and making customization quite practical and available to people.”
Read Our Manufacturing Blog: Incorporating AM in Industrial Operations
As the pandemic has continued, additive manufacturing has proven to be a viable solution for time-sensitive production. If the dental industry can serve as an example, it can become a solution for on-demand production. So, what are the next steps to create a wholly distributed and net-zero manufacturing network?
While the additive manufacturing industry has seen many successes during the pandemic, it has not been without some setbacks. The aerospace and automotive industry has seen significant losses during the pandemic. The lack of new business has hurt the industry: GE has had to cut 10,000 aviation jobs, Boeing has slashed aircraft production, and Airbus reports a 55 percent loss of revenue. For both sectors, additive manufacturing is used to produce hardware components, particularly metal 3D printed parts.
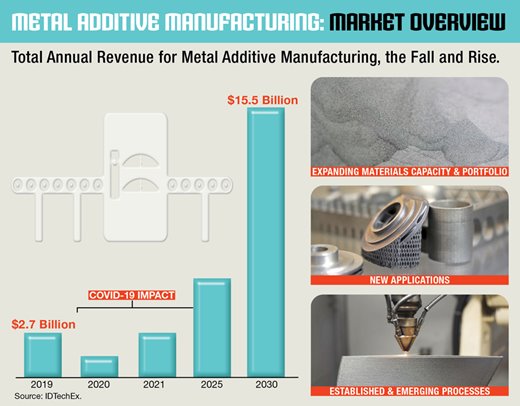
Richard Collins, a principal analyst at IDTechEx in Cambridge, England, spoke about how the metal additive manufacturing industry will get worse before it gets better. IDTechEx recently released the Metal Additive Manufacturing 2020-2030 report recapping the trends of 2020 and the future of additive manufacturing over the next decade. Within the report, IDTechEx highlighted how printing costs are lowering as new powder and feedstock variants are being introduced into the market. The gradual decrease in material costs, the maturity of the supply chain, and the increase in competition will foster a larger marketplace for additive manufacturing.
While the company reports how COVID-19 negatively impacted metal additive manufacturing, the effect is expected to be short-term. The pandemic has forced companies to invest in the future of additive manufacturing. An increased number of market players will innovative novel processes and materials to facilitate easier production of additive manufacturing parts. There was notable activity between 2017-2019 in metal powder for additive manufacturing; this was predominantly in the aerospace industry from the likes of Carpenter, GKN, and AP&C, a GE Additive company. There has remained a large amount of material development in 2020, which will continue to expand in the upcoming decade.
“There’s been a negative impact, but we have started to look forward and reevaluate how people view the distributed supply chain,” Collins said. “If companies are seriously considering adopting a distributed supply chain, additive manufacturing is a key partner.”
The motivation behind distributed, 3D net-zero manufacturing is to address the challenges of global social, economic, and ecological trends, all of which were highlighted during the pandemic. Success requires systemic perspective and consideration of 3D additive manufacturing as a cyberphysical solution built by integrating operating technologies (OT) and information technologies (IT). In a 3D digital manufacturing environment—the 3D-printing devices, power, and support equipment—are the OT, and data gathering devices and computing equipment are the IT.
“There is a range of 3D technologies that are available to go from art to part,” said Chandrakant Patel, chief engineer at HP and an ASME fellow. “By building microgrids of manufacturing, you don’t need to build a gigantic plant. When you have modular factories globally distributed at scale and powered by local sources of renewable energy, such as solar and even waste streams (methane from anaerobic digestion of manure from dairy cows as an example), you can now drive toward net-zero manufacturing.”
Listen to ASME TechCast: Additive Manufacturing Revolutionizes Aerospace Design
In the December special issue of The Bridge for the National Academy of Engineering, Patel wrote, “[in] 3D digital manufacturing, the OT is made up of 3D printers, and the IT ‘art-to-part’ cyberphysical pipeline starts with part design anywhere in the world. Then it is the transfer of digital data to print parts in a given locality and delivery to the customer. The need-based provisioning capacity of 3D digital manufacturing [during the pandemic] has shown that it can enable resilient supply chains.”
According to Patel, 3D digital manufacturing’s success requires a holistic perspective that encompasses design, devices, and the digital factory. When it comes to design, the designer can take advantage of the ability of 3D printing to create shapes and contours not previously possible with the appropriate choice of 3D printing technology. With a range of additive manufacturing printers now available, designers and creators need to rethink how to manufacture parts. The model of buying parts in surplus must shift to smaller batches of highly custom parts. Like the dental fixtures, these parts will fit precisely the project’s needs—requiring precision and fine-tuned design from the engineer.
With respect to the 3D printer devices and the digital factory, large amounts of machine-generated data are available to deliver accurate and expeditious outcomes.
“However, in this context, treating the 3D printer device as a black box with the assumption that large amounts of data from the printer and AI alone will create the accurate outcomes is incorrect,” Patel said. “Deep domain understanding of physical and material sciences is fundamental to a successful model of operating the 3D art-to-part pipeline at the intersection of the domain, data, and AI.”
There are still hurdles to clear before distributed manufacturing can be achieved, regulations being the tallest. The pandemic is still ongoing, but several supply chains have returned to normal production. During the early months of the pandemic, the progress made was highly incentivized by the EUAs granted by the FDA. According to Wilczynski, the healthcare emergency was a significant catalyst for the industry.
“As soon as the EUAs expire, any component made through non-conventional means is no longer allowed to be used. There’s an assumption that the current series of regulations and requirements in place is what’s best for the industry,” Wilczynski said.
However, as Remacha and her team discovered, several of the regulations did not apply in an additive manufacturing setting.
“We were not familiar with the culture of regulations within the medical industry. We were calling the agency to ensure they had all the information, and it took a long time for them to review the documentation,” Remacha said. “In the end, the agency found areas where the requirements did not make sense [for additive parts], and for the following parts produced, it was easier to certify.”
With the changes in regulations, the creation of new materials, and the lowering price point for 3D printers, additive manufacturing is becoming more feasible.
“There is a cutoff point for various technologies. Based on how many parts you need and the customization required, 3D is more cost-effective,” Patel said. “But if you say that the world is shifting toward a highly personalized, customized, individualized, and on-demand distributed manufacturing model, which is sustainable, we will see the world move toward additive manufacturing.”
Carlos M. González is special projects manager.
Join ASME’s AM Tech Forum Celebrating AM Innovation
At a networking event at a local hospital in March 2020, a physician colleague of Remacha’s warned her about how COVID-19 was ramping up. More than 1,200 cases had been confirmed since the first case on January 31—barely over a month earlier. The doctor suggested that everyone should work from home and go remote. Sure enough, Spain enacted a lockdown the very next week.
What happened next in Spain became a reoccurring theme all over the world. Supply chains were disrupted, resulting in a lack of goods for consumers and raw materials for industry. Hospitals were flooded with COVID-19 cases, and doctors were forced to choose which patients would get a ventilator because of limited supply. Healthcare workers and the general public alike scrambled for personal protective equipment. It was a nightmare.
Remacha and her team soon started to receive requests from local hospitals to 3D print equipment. Juan Angel Muñoz López, a nursing supervisor at Príncipe de Asturias Hospital in Alcalá de Henares just outside of Madrid, reached out to Barcelona’s 3D Printing and Digital Manufacturing Center of Excellence. “He told us they had already reached the point where they had to decide who lived and who didn’t. They didn’t have enough respirators for everyone,” Remacha said. Muñoz wanted HP’s help to fabricate a part the ICU nursing staff had designed that could connect multiple standard elements to create a continuous positive airway pressure (CPAP) device. The CPAP device could help increase a patients’ blood oxygen levels above 90 percent, potentially sparing them from using a ventilator.
Within two days, Remacha and the HP team had the design evaluated, printed, and sent back to the hospital for validation. And within just two weeks, the CPAP cross-connector device was approved by the Agencia Española de Medicamentos y Productos Sanitarios, the Spanish medical device regulator.
Stories like these threw a spotlight on additive manufacturing and its potential for fast, flexible production of critically needed parts. Whether it was face shields, nasopharyngeal swabs, or respiratory masks, additive manufacturing rose to the challenge to fill in the gap caused by a disrupted supply chain. Now, some industry observers are starting to ask whether 3D printing could be the future of manufacturing.
Additive Manufacturing Takes Center Stage
Disrupted supply chains led several hospitals to ask additive manufacturing companies for help by producing potentially life-saving equipment. For companies like HP, it was a challenge on how to begin.
“We had no idea what was needed,” Remacha said. “We design 3D printers. We don’t design medical devices.”
HP’s first step was to set up an online form through which customers and users could submit requests for 3D-printed accessories to help them battle COVID-19 in their community. At first, people asked for hands-free door openers and mask adjusters. As the pandemic went on, healthcare workers started to ask for face shields. Remacha and her team had a virtual meeting with one of the doctors they work with to formalize the design on how to construct a face shield properly.
“We didn’t know what requirements we had to meet. The healthcare workers were now working 12-hour shifts, and they told us that comfort was one of the most important factors,” Remacha said.
The HP team met on a Saturday and by Monday had designed and printed a face shield prototype with an adjustable band. The team used taxis to ship parts back and forth between the hospital to ensure fit and functionality. By the following Friday, the design was on its fourth iteration. The design received full certification based on the European EN-166 standard a few weeks later.
Recommended for You: Lakshmi Vendra Reviews AM’s Use in Oil and Gas
Since announcing their efforts to help the medical community, HP and its global community partners produced more than 6 million parts for medical applications.
The ease of regulations and standards aided the additive manufacturing industry significantly during the pandemic. In the U.S., the Food and Drug Administration granted emergency use authorization (EUA) to 3D print parts for medical applications. For instance, Somerville, Mass.-based Formlabs was given an EUA to print and ship bi-level positive airway pressure (BiPAP) adapters to hospitals throughout the U.S. to combat ventilator shortages at the beginning of the pandemic.
America Makes is one of the nation’s leading and collaborative partners in additive manufacturing and 3D printing technology research and innovation. At the very start of the pandemic, America Makes, along with their partner network, documented a series of issues surrounding the supply chain and presented those to the commissioner of the FDA.
“Within days of the pandemic starting, we told the FDA that the AM community is ready to help. They’re sitting on the sidelines and can’t get engaged because they don’t know answers to these various [regulatory] questions,” said John Wilczynski, executive director at America Makes. “Almost immediately, the formation of an interagency agreement, between the Veterans Health Administration (VA) office, the FDA, and the National Institute of Health (NIH), all started to work together. There was a tremendous flow of information that was disseminated to the AM community, which helped manufacturers get started.”
On-Demand Manufacturing Today
The pandemic has encouraged hospitals to consider on-premise or on-demand manufacturing. Systems like the VA have taken significant strides in providing point of care device manufacturing, such as on-premise 3D printing. According to Wilczynski, the pandemic presents an opportunity to reconsider the supply chain and how parts are manufactured.
“In most cases, people haven’t gone down that path,” Wilczynski said. “The procurement system associated with healthcare does not provide the freedom to buy a single $50 item, which normally costs $1 for daily use, but with AM, we can. It’s an interesting idea because we are shifting from a consumable approach to a reusable approach.”
3D Systems in Rock Hill, S.C., partnered with the VA to design medical devices and take them through FDA clearance. The two organizations will use AM solutions to provide point of care, next-generation medical devices.
The initiative spurred out of the initial collaboration to address the lack of face masks within the VA hospitals at the start of the pandemic.
The next design was to develop a 3D-printed nasopharyngeal (NP) swab that could be manufactured on production-level equipment. The shortage of NP swabs was a significant hurdle as large-scale testing began to ramp up. 3D Systems were able to test and validate a custom medical-grade NP swab design out of DuraFrom ProxX PA using its ProX SLS 6100 printer. The material is a strong, tough thermoplastic material suitable for select medical devices such as NP swabs. The company was able to produce 3,000 swabs every 24 hours per printer.
“What began during the pandemic in response to a critical need has expanded to change the way healthcare is delivered,” said Menno Ellis, executive vice president of healthcare solutions at 3D Systems. “This is a one-of-a-kind collaboration between an additive manufacturing solutions provider and one of the world’s largest integrated health care systems to accelerate innovation in the medical device production and deployment arena.”
3D Systems has had plenty of success in delivering on-demand medical additive manufacturing applications within the dental industry. Dental appliances, including crowns, bridges, and dentures, are produced using additive manufacturing at a high volume and on-demand. For example, the dentist would take an intraoral scan and create a digital impression that is instantly analyzed. If a 3D printer is on-site, the dentist could manufacturer a try-in mold the very same day and test it on the patient. The patient takes the mold home to try for a few days before finalizing the dental fixture. The entire process cuts down what used to take weeks into a matter of days.
“We are seeing increasing use of patient-specific instruments and medical devices, and expect this will continue to accelerate,” Ellis said. “This is where 3D printing shines and differentiates itself from other technologies, because you have the benefit of producing in units of one and making customization quite practical and available to people.”
Read Our Manufacturing Blog: Incorporating AM in Industrial Operations
As the pandemic has continued, additive manufacturing has proven to be a viable solution for time-sensitive production. If the dental industry can serve as an example, it can become a solution for on-demand production. So, what are the next steps to create a wholly distributed and net-zero manufacturing network?
Distributed and Net-Zero Manufacturing
While the additive manufacturing industry has seen many successes during the pandemic, it has not been without some setbacks. The aerospace and automotive industry has seen significant losses during the pandemic. The lack of new business has hurt the industry: GE has had to cut 10,000 aviation jobs, Boeing has slashed aircraft production, and Airbus reports a 55 percent loss of revenue. For both sectors, additive manufacturing is used to produce hardware components, particularly metal 3D printed parts.
Richard Collins, a principal analyst at IDTechEx in Cambridge, England, spoke about how the metal additive manufacturing industry will get worse before it gets better. IDTechEx recently released the Metal Additive Manufacturing 2020-2030 report recapping the trends of 2020 and the future of additive manufacturing over the next decade. Within the report, IDTechEx highlighted how printing costs are lowering as new powder and feedstock variants are being introduced into the market. The gradual decrease in material costs, the maturity of the supply chain, and the increase in competition will foster a larger marketplace for additive manufacturing.
While the company reports how COVID-19 negatively impacted metal additive manufacturing, the effect is expected to be short-term. The pandemic has forced companies to invest in the future of additive manufacturing. An increased number of market players will innovative novel processes and materials to facilitate easier production of additive manufacturing parts. There was notable activity between 2017-2019 in metal powder for additive manufacturing; this was predominantly in the aerospace industry from the likes of Carpenter, GKN, and AP&C, a GE Additive company. There has remained a large amount of material development in 2020, which will continue to expand in the upcoming decade.
“There’s been a negative impact, but we have started to look forward and reevaluate how people view the distributed supply chain,” Collins said. “If companies are seriously considering adopting a distributed supply chain, additive manufacturing is a key partner.”
The motivation behind distributed, 3D net-zero manufacturing is to address the challenges of global social, economic, and ecological trends, all of which were highlighted during the pandemic. Success requires systemic perspective and consideration of 3D additive manufacturing as a cyberphysical solution built by integrating operating technologies (OT) and information technologies (IT). In a 3D digital manufacturing environment—the 3D-printing devices, power, and support equipment—are the OT, and data gathering devices and computing equipment are the IT.
“There is a range of 3D technologies that are available to go from art to part,” said Chandrakant Patel, chief engineer at HP and an ASME fellow. “By building microgrids of manufacturing, you don’t need to build a gigantic plant. When you have modular factories globally distributed at scale and powered by local sources of renewable energy, such as solar and even waste streams (methane from anaerobic digestion of manure from dairy cows as an example), you can now drive toward net-zero manufacturing.”
Listen to ASME TechCast: Additive Manufacturing Revolutionizes Aerospace Design
In the December special issue of The Bridge for the National Academy of Engineering, Patel wrote, “[in] 3D digital manufacturing, the OT is made up of 3D printers, and the IT ‘art-to-part’ cyberphysical pipeline starts with part design anywhere in the world. Then it is the transfer of digital data to print parts in a given locality and delivery to the customer. The need-based provisioning capacity of 3D digital manufacturing [during the pandemic] has shown that it can enable resilient supply chains.”
According to Patel, 3D digital manufacturing’s success requires a holistic perspective that encompasses design, devices, and the digital factory. When it comes to design, the designer can take advantage of the ability of 3D printing to create shapes and contours not previously possible with the appropriate choice of 3D printing technology. With a range of additive manufacturing printers now available, designers and creators need to rethink how to manufacture parts. The model of buying parts in surplus must shift to smaller batches of highly custom parts. Like the dental fixtures, these parts will fit precisely the project’s needs—requiring precision and fine-tuned design from the engineer.
With respect to the 3D printer devices and the digital factory, large amounts of machine-generated data are available to deliver accurate and expeditious outcomes.
“However, in this context, treating the 3D printer device as a black box with the assumption that large amounts of data from the printer and AI alone will create the accurate outcomes is incorrect,” Patel said. “Deep domain understanding of physical and material sciences is fundamental to a successful model of operating the 3D art-to-part pipeline at the intersection of the domain, data, and AI.”
The World is Shifting
There are still hurdles to clear before distributed manufacturing can be achieved, regulations being the tallest. The pandemic is still ongoing, but several supply chains have returned to normal production. During the early months of the pandemic, the progress made was highly incentivized by the EUAs granted by the FDA. According to Wilczynski, the healthcare emergency was a significant catalyst for the industry.
“As soon as the EUAs expire, any component made through non-conventional means is no longer allowed to be used. There’s an assumption that the current series of regulations and requirements in place is what’s best for the industry,” Wilczynski said.
However, as Remacha and her team discovered, several of the regulations did not apply in an additive manufacturing setting.
“We were not familiar with the culture of regulations within the medical industry. We were calling the agency to ensure they had all the information, and it took a long time for them to review the documentation,” Remacha said. “In the end, the agency found areas where the requirements did not make sense [for additive parts], and for the following parts produced, it was easier to certify.”
With the changes in regulations, the creation of new materials, and the lowering price point for 3D printers, additive manufacturing is becoming more feasible.
“There is a cutoff point for various technologies. Based on how many parts you need and the customization required, 3D is more cost-effective,” Patel said. “But if you say that the world is shifting toward a highly personalized, customized, individualized, and on-demand distributed manufacturing model, which is sustainable, we will see the world move toward additive manufacturing.”
Carlos M. González is special projects manager.
Join ASME’s AM Tech Forum Celebrating AM Innovation