Government Action Creates a Ripple Effect on Supply Chain
Government Action Creates a Ripple Effect on Supply Chain
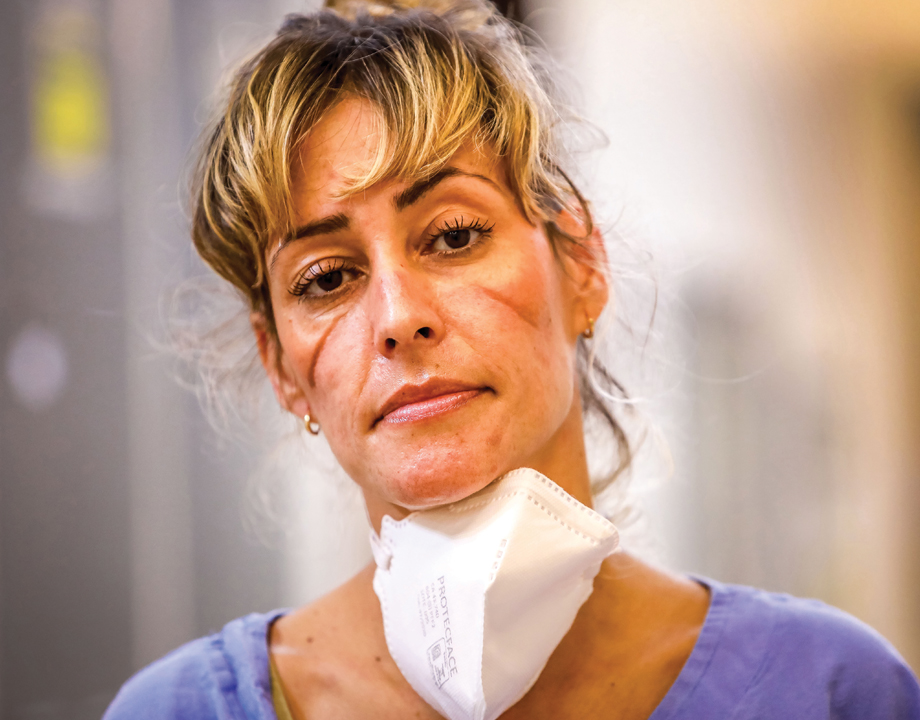

Federal action helped relieve medical supply-chain issues caused by the COVID-19 pandemic, but the actions have impacted the biomedical industry in unexpected ways. Image: Getty
Some of the images from the first weeks of the COVID-19 pandemic are indelible: doctors and nurses wearing garbage bags instead of gowns and reusing the same protective mask for days at a time; homemade plastic face shields replacing professional-grade models; live coverage of deliveries of personal protective equipment to hard-hit hospitals. The haunting photo of the nurse shown above was taken in those uncertain weeks.
The medical supply chain was so hard hit during the spring of 2020 that hospitals and even state governments would arrange for delivery of PPE in secret, concerned that vital supplies would be commandeered.
When the COVID-19 pandemic first hit, it impacted supply chains across all industries and sectors. Fortunately, the days of healthcare workers wearing plastic bags in lieu of proper medical gear are behind us. But as global economies adjusted and began to regain their equilibrium, the biomedical engineering sector found itself lagging other sectors in returning to normal operations. Two key factors impacted this return to normalcy: Quality control issues in manufacturing were brought on due to a dearth of workers and supplies, and the Defense Production Act—the very measure that helped restock hospitals with the PPEs they need—created scarcity in other parts of the supply chain.
More on Biomedical Technology: Metamaterials Expand Possibilities in Biomedical Engineering
As we enter the third year of the COVID era, it is crucial that policymakers and manufacturers revisit the medical supply chain. We all need to better understand how the Defense Production Act can distort the supply chain in ways that ripple out beyond the specific productions being requisitioned. More importantly, the country needs to commit itself to a domestic manufacturing capability so that it won’t be left with critical shortages the next time a crisis hits.
The Defense Production Act (DPA) was passed in 1950 at start of Korean War. The legislation was drafted to provide the president of the United States the authority to utilize domestic industries to assist the economy and the country at-large in times of crisis. At its inception, this authority included the ability to set wages for workers in factories, and the corresponding price of goods produced. The DPA has evolved over time (the most recent reauthorization was in 2019) and its authority can also be used to mandate U.S.-based companies to pivot and manufacture essential goods that are in short supply. The current iteration of DPA grants the president the authority to “allocate materials, services, and facilities” for national defense purposes, and take actions to restrict hoarding of needed supplies. Companies that fail to comply with the DPA mandate can be found guilty of a crime and face fines of up $10,000.
The original impetus for the DPA was to provide reliable production of materials and goods critical to warfighting, such as steel and aluminum for tanks and planes or oil for transportation. Over time, the act was used for new technologies and materials useful for defense—everything from silicon carbide ceramics to radiation-hardened microelectronics. Since 2020, both Presidents Trump and Biden have enacted the DPA as part of their efforts to help the U.S. better respond to the COVID-19 pandemic and overcome the supply chain and manufacturing constraints that have arisen.
For instance, President Trump used the DPA to mandate General Motors to leverage its manufacturing facilities and acumen to start producing ventilators to fill the acute need that arose in Spring 2020. Also during this time, in efforts to address the shortage of PPE, President Trump directed Minnesota-based manufacturer 3M to reduce the exports of its domestically manufactured N95 masks and preferentially send more of the masks the company produced in its Singapore facility to the U.S.
President Biden has also invoked the DPA to help the U.S. by seeking to improve supply chains and expand the national stockpile of essential goods. In one of his first acts as president, he ordered a 100-day review of all U.S. supply chains; that review identified four key areas of prioritization: semiconductors; large capacity batteries; critical minerals and materials; and pharmaceuticals and active pharmaceutical ingredients. Subsequently, from those four categories, President Biden has so far issued 18 priority ratings for U.S. companies to assist in mitigating these supply-chain constraints and increasing the national stockpile of essential goods.
Take the Quiz: The History of Artificial Knees
The authority of the DPA is a powerful tool, and presidents must be careful to wield it in a nuanced manner. The U.S. economy is strongly tied to international trade, and American manufacturers are dependent on global supply chains. Using the DPA to improve the situation at home must be balanced with reducing the potential for long-term mistrust by our international trading partners.
When the Trump administration ordered 3M, a U.S.-based company, to stop exporting an essential good to global partners, it ran the risk of in fact reducing the country’s already constricted mask supply chain. That’s because 3M was not the only producer of N95 masks; companies located in other countries were also producing and exporting N95 masks to the U.S. Restricting all 3M-produced masks to the U.S. risked aggravating those other trading partners and spurring them to cease export of their N95 masks to the U.S.
The impact of DPA mandates can be felt long after the immediate supply crisis is over. Earlier invocations of the act were designed to command production to help create a long-term domestic supply of critical materials. There is a growing concern, however, that the way these mandates have been implemented during the COVID-19 pandemic could have a negative domino effect on the biomedical engineering sector here in the U.S.
When Presidents Trump and Biden mandated key manufacturers to pivot and respond to the COVID-19 pandemic, for instance, not only manufacturers but also key suppliers had to pivot and prioritize the DPA-mandated pandemic response. This left medical device companies and research labs scrambling for replacement suppliers to continue production and research of non-COVID diseases. The fear is that this disruption could constrain supply of non-COVID biomedical devices or increase their cost.
The implementation of the DPA also raises a business continuity issue. With COVID-19 mitigation measures currently serving as the principal priority for those government-mandated companies, it is tough for companies not directly fighting COVID to build up supplies. Since the start of the pandemic, materials such as stainless steel, silicon microfluidics, pipettes, and small-to-medium sized gloves have at times been challenging to track down, along with the highly discussed lack of general PPE that had long been used in lab and hospital settings prior to COVID-19. If companies and researchers are unable to obtain the materials needed to continue operations, the likelihood of business constrictions is very high.
Other long-term effects could take decades to be fully felt. Due to the difficulties obtaining requisite media, a significant amount of biomedical research in non-COVID-19 areas such as stem cells has been halted or drastically pushed back. This is cause for concern because it is delaying potential life-changing treatments, therapies, and advances in research that will extend beyond the end of this pandemic.
Research delays also mean that many research grants are coming to an end with unspent funds and no option for cost extension, and researchers are not performing as expected on grants due to personal and professional setbacks. If the importance of delays is not recognized, such postponements all could have significant impacts down the line for the biomedical and healthcare fields.
Another issue compounding the shortages arising due to the DPA is the issue of quality control. This is especially important in the biomedical field, where manufacturing tolerances are tight and the industry comes under the jurisdiction of the U.S. Food and Drug Administration, which is widely acknowledged as strict.
The experience with pathogen-filtering masks is illustrative. Since the onset of the pandemic, N95 masks have been periodically challenging to obtain for both frontline workers and the general public. Cloth masks were deemed suitable for the general public until late 2021, when the Centers for Disease Control (CDC) suggested that such masks did not protect against the spread of new COVID-19 variants. That led to a risk of the U.S. once more seeing a run N95 and other filtered masks. The rapid increase in demand over a short period of time leaves domestic manufacturers struggling to catch up, while many imported masks have been failing FDA quality control tests. This left the imports unable to be distributed.
In response to these severe shortages, many biomedical lab researchers started using KF94 masks instead of N95 or KN95 masks. These masks are manufactured in South Korea and certified by the South Korean government to meet a standard similar to the U.S. N95 and Chinese KN95 standards. Additionally, KF94 masks come individually packaged, which cuts down on fraudulent resellers and additional quality-control concerns.
Another avenue for increased mask supply was manufactured by new entries into the domestic market. In 2020, several U.S. companies started manufacturing their own N95 masks. Unfortunately, many of these masks also failed to obtain FDA approval due to quality-control concerns. As the local manufacturing community sought to alleviate the dearth of PPE in the U.S. through new manufacturing capabilities such as 3D printing, this raised a new challenge for the FDA and other regulatory bodies: How could they speed their process for determining what new materials are safe and effective for mask production?
It has been two years since the start of the COVID-19 pandemic and the ensuing initial supply constraints, but the biomedical industry continues to face major supply shortages brought on by quality-control issues.
In addition to masks, nitrile gloves continue to be difficult to obtain, and these shortages are compounded by black-market sellers peddling used gloves that they repackage, causing major safety concerns. Prior to the COVID-19 pandemic, nitrile gloves had a very low defect rate, but in part due to unscrupulous reseller policies and difficulty obtaining the requisite materials, the defect rate of gloves has now increased exponentially, with some biomedical researchers stating they are forced to throw out as many as 10 percent of their gloves due to rips and other defects.
A recent ASME task force in which we were involved looked at the experience of the past two years and the impact of the Defense Production Act on the biomedical supply chain. We saw some steps that could improve the situation and reduce some of the unintended effects that are rippling through the industry.
First, the U.S. needs to strengthen its domestic manufacturing infrastructure. The enactment of the DPA highlighted the inability of the manufacturing sector to rapidly scale up in a time of crisis, which put biomedical manufacturers and suppliers in a position where they were unable to respect the DPA mandate while simultaneously fulfilling their non-COVID manufacturing obligations.
One step that must be taken is creating short, duplicative supply chains to ensure that in future, the U.S. will not be put in a position of need once more. It is also important to revisit stockpiling inventory to avoid shortages of necessary equipment and supplies when crises hit.
In addition, federal agencies must better understand the weight and significance of the DPA and the role the government overall can play in supporting industry, so it can honor its DPA obligations.
Small steps in this direction have begun. The Government Accountability Office stated in a December 2021 report that some federal agencies had started to take measures to improve DPA understanding. The report notes that the Department of Human Health and Services, which houses agencies such as the National Institutes of Health, Food and Drug Administration, and Centers for Disease Control and Prevention, had established a DPA-specific office within the Office of the Assistant Secretary for Preparedness and Response in June 2020. The Department of Defense also established an office to support interagency acquisition efforts in October 2020.
Bioengineering Blog: Supply Chains and the Health Crisis
Those steps are a good start but insufficient. Moving forward, the government needs to leverage its multilateral hegemony to inform and provide guidance to the biomedical industry on how to respond to the DPA mandates in a clear and targeted manner that does not have a disruptive domino effect downstream.
Finally, public-private partnerships can be better leveraged to encourage expertise and knowledge across sectors. With quality control continuing to be an issue, partnering with industry to understand the most important materials and functions of essential goods will be key to agility in the future. As we have seen with the establishment of the Manufacturing USA network and the biomedical-specific institutes such as ARMI, NIIMBL, and BioMADE, working closely across sectors facilitates the sharing and implementation of best practices, standards, and guidance to create a nimbler infrastructure that can withstand and adjust to supply-chain constraints.
COVID-19 has left no industry or activity untouched, but the pandemic continues to negatively impact certain sectors more than others. Despite the importance of the research, development, and products coming out of the biomedical sector, policy decisions continue to have far-reaching implications on the sector’s ability to bounce back.
Though much has been done to create a “new normal,” there is still a way to go. It will take a thoughtful, considered response to ensure the biomedical sector can produce the results we all rely on.
Kevin D. Costa is the director of cardiovascular cell and tissue engineering at the Cardiovascular Research Institute of the Icahn School of Medicine at Mount Sinai in New York City. Costa was also the chair of the ASME Bioengineering Public Policy Task Force (BPPTF). Charles Y. Lee is an ASME Fellow and the director of the Center for Biomedical Engineering and Science at the University of North Carolina at Charlotte. Lee was the ASME Committee on Government Relations Liaison to the BPPTF.
The authors would like to acknowledge the other members of the task force for their contributions: Martin L. Tanaka, P.E., of Western Carolina University in Cullowhee, N.C.; Joe Budzinski, director of the Global Development Center at Johnson & Johnson; Roger Narayan of North Carolina State University in Raleigh; C. Ross Ethier, chair in bioengineering at the Georgia Institute of Technology and the Emory University School of Medicine; and Mike Hooven, CEO at Enable Injections.
The medical supply chain was so hard hit during the spring of 2020 that hospitals and even state governments would arrange for delivery of PPE in secret, concerned that vital supplies would be commandeered.
When the COVID-19 pandemic first hit, it impacted supply chains across all industries and sectors. Fortunately, the days of healthcare workers wearing plastic bags in lieu of proper medical gear are behind us. But as global economies adjusted and began to regain their equilibrium, the biomedical engineering sector found itself lagging other sectors in returning to normal operations. Two key factors impacted this return to normalcy: Quality control issues in manufacturing were brought on due to a dearth of workers and supplies, and the Defense Production Act—the very measure that helped restock hospitals with the PPEs they need—created scarcity in other parts of the supply chain.
More on Biomedical Technology: Metamaterials Expand Possibilities in Biomedical Engineering
As we enter the third year of the COVID era, it is crucial that policymakers and manufacturers revisit the medical supply chain. We all need to better understand how the Defense Production Act can distort the supply chain in ways that ripple out beyond the specific productions being requisitioned. More importantly, the country needs to commit itself to a domestic manufacturing capability so that it won’t be left with critical shortages the next time a crisis hits.
Imposing Order
The Defense Production Act (DPA) was passed in 1950 at start of Korean War. The legislation was drafted to provide the president of the United States the authority to utilize domestic industries to assist the economy and the country at-large in times of crisis. At its inception, this authority included the ability to set wages for workers in factories, and the corresponding price of goods produced. The DPA has evolved over time (the most recent reauthorization was in 2019) and its authority can also be used to mandate U.S.-based companies to pivot and manufacture essential goods that are in short supply. The current iteration of DPA grants the president the authority to “allocate materials, services, and facilities” for national defense purposes, and take actions to restrict hoarding of needed supplies. Companies that fail to comply with the DPA mandate can be found guilty of a crime and face fines of up $10,000.
The original impetus for the DPA was to provide reliable production of materials and goods critical to warfighting, such as steel and aluminum for tanks and planes or oil for transportation. Over time, the act was used for new technologies and materials useful for defense—everything from silicon carbide ceramics to radiation-hardened microelectronics. Since 2020, both Presidents Trump and Biden have enacted the DPA as part of their efforts to help the U.S. better respond to the COVID-19 pandemic and overcome the supply chain and manufacturing constraints that have arisen.
For instance, President Trump used the DPA to mandate General Motors to leverage its manufacturing facilities and acumen to start producing ventilators to fill the acute need that arose in Spring 2020. Also during this time, in efforts to address the shortage of PPE, President Trump directed Minnesota-based manufacturer 3M to reduce the exports of its domestically manufactured N95 masks and preferentially send more of the masks the company produced in its Singapore facility to the U.S.
President Biden has also invoked the DPA to help the U.S. by seeking to improve supply chains and expand the national stockpile of essential goods. In one of his first acts as president, he ordered a 100-day review of all U.S. supply chains; that review identified four key areas of prioritization: semiconductors; large capacity batteries; critical minerals and materials; and pharmaceuticals and active pharmaceutical ingredients. Subsequently, from those four categories, President Biden has so far issued 18 priority ratings for U.S. companies to assist in mitigating these supply-chain constraints and increasing the national stockpile of essential goods.
Take the Quiz: The History of Artificial Knees
The authority of the DPA is a powerful tool, and presidents must be careful to wield it in a nuanced manner. The U.S. economy is strongly tied to international trade, and American manufacturers are dependent on global supply chains. Using the DPA to improve the situation at home must be balanced with reducing the potential for long-term mistrust by our international trading partners.
When the Trump administration ordered 3M, a U.S.-based company, to stop exporting an essential good to global partners, it ran the risk of in fact reducing the country’s already constricted mask supply chain. That’s because 3M was not the only producer of N95 masks; companies located in other countries were also producing and exporting N95 masks to the U.S. Restricting all 3M-produced masks to the U.S. risked aggravating those other trading partners and spurring them to cease export of their N95 masks to the U.S.
The impact of DPA mandates can be felt long after the immediate supply crisis is over. Earlier invocations of the act were designed to command production to help create a long-term domestic supply of critical materials. There is a growing concern, however, that the way these mandates have been implemented during the COVID-19 pandemic could have a negative domino effect on the biomedical engineering sector here in the U.S.
When Presidents Trump and Biden mandated key manufacturers to pivot and respond to the COVID-19 pandemic, for instance, not only manufacturers but also key suppliers had to pivot and prioritize the DPA-mandated pandemic response. This left medical device companies and research labs scrambling for replacement suppliers to continue production and research of non-COVID diseases. The fear is that this disruption could constrain supply of non-COVID biomedical devices or increase their cost.
The implementation of the DPA also raises a business continuity issue. With COVID-19 mitigation measures currently serving as the principal priority for those government-mandated companies, it is tough for companies not directly fighting COVID to build up supplies. Since the start of the pandemic, materials such as stainless steel, silicon microfluidics, pipettes, and small-to-medium sized gloves have at times been challenging to track down, along with the highly discussed lack of general PPE that had long been used in lab and hospital settings prior to COVID-19. If companies and researchers are unable to obtain the materials needed to continue operations, the likelihood of business constrictions is very high.
Other long-term effects could take decades to be fully felt. Due to the difficulties obtaining requisite media, a significant amount of biomedical research in non-COVID-19 areas such as stem cells has been halted or drastically pushed back. This is cause for concern because it is delaying potential life-changing treatments, therapies, and advances in research that will extend beyond the end of this pandemic.
Research delays also mean that many research grants are coming to an end with unspent funds and no option for cost extension, and researchers are not performing as expected on grants due to personal and professional setbacks. If the importance of delays is not recognized, such postponements all could have significant impacts down the line for the biomedical and healthcare fields.
Quality Control
Another issue compounding the shortages arising due to the DPA is the issue of quality control. This is especially important in the biomedical field, where manufacturing tolerances are tight and the industry comes under the jurisdiction of the U.S. Food and Drug Administration, which is widely acknowledged as strict.
The experience with pathogen-filtering masks is illustrative. Since the onset of the pandemic, N95 masks have been periodically challenging to obtain for both frontline workers and the general public. Cloth masks were deemed suitable for the general public until late 2021, when the Centers for Disease Control (CDC) suggested that such masks did not protect against the spread of new COVID-19 variants. That led to a risk of the U.S. once more seeing a run N95 and other filtered masks. The rapid increase in demand over a short period of time leaves domestic manufacturers struggling to catch up, while many imported masks have been failing FDA quality control tests. This left the imports unable to be distributed.
In response to these severe shortages, many biomedical lab researchers started using KF94 masks instead of N95 or KN95 masks. These masks are manufactured in South Korea and certified by the South Korean government to meet a standard similar to the U.S. N95 and Chinese KN95 standards. Additionally, KF94 masks come individually packaged, which cuts down on fraudulent resellers and additional quality-control concerns.
Another avenue for increased mask supply was manufactured by new entries into the domestic market. In 2020, several U.S. companies started manufacturing their own N95 masks. Unfortunately, many of these masks also failed to obtain FDA approval due to quality-control concerns. As the local manufacturing community sought to alleviate the dearth of PPE in the U.S. through new manufacturing capabilities such as 3D printing, this raised a new challenge for the FDA and other regulatory bodies: How could they speed their process for determining what new materials are safe and effective for mask production?
It has been two years since the start of the COVID-19 pandemic and the ensuing initial supply constraints, but the biomedical industry continues to face major supply shortages brought on by quality-control issues.
In addition to masks, nitrile gloves continue to be difficult to obtain, and these shortages are compounded by black-market sellers peddling used gloves that they repackage, causing major safety concerns. Prior to the COVID-19 pandemic, nitrile gloves had a very low defect rate, but in part due to unscrupulous reseller policies and difficulty obtaining the requisite materials, the defect rate of gloves has now increased exponentially, with some biomedical researchers stating they are forced to throw out as many as 10 percent of their gloves due to rips and other defects.
Finding a Cure
A recent ASME task force in which we were involved looked at the experience of the past two years and the impact of the Defense Production Act on the biomedical supply chain. We saw some steps that could improve the situation and reduce some of the unintended effects that are rippling through the industry.
First, the U.S. needs to strengthen its domestic manufacturing infrastructure. The enactment of the DPA highlighted the inability of the manufacturing sector to rapidly scale up in a time of crisis, which put biomedical manufacturers and suppliers in a position where they were unable to respect the DPA mandate while simultaneously fulfilling their non-COVID manufacturing obligations.
One step that must be taken is creating short, duplicative supply chains to ensure that in future, the U.S. will not be put in a position of need once more. It is also important to revisit stockpiling inventory to avoid shortages of necessary equipment and supplies when crises hit.
In addition, federal agencies must better understand the weight and significance of the DPA and the role the government overall can play in supporting industry, so it can honor its DPA obligations.
Small steps in this direction have begun. The Government Accountability Office stated in a December 2021 report that some federal agencies had started to take measures to improve DPA understanding. The report notes that the Department of Human Health and Services, which houses agencies such as the National Institutes of Health, Food and Drug Administration, and Centers for Disease Control and Prevention, had established a DPA-specific office within the Office of the Assistant Secretary for Preparedness and Response in June 2020. The Department of Defense also established an office to support interagency acquisition efforts in October 2020.
Bioengineering Blog: Supply Chains and the Health Crisis
Those steps are a good start but insufficient. Moving forward, the government needs to leverage its multilateral hegemony to inform and provide guidance to the biomedical industry on how to respond to the DPA mandates in a clear and targeted manner that does not have a disruptive domino effect downstream.
Finally, public-private partnerships can be better leveraged to encourage expertise and knowledge across sectors. With quality control continuing to be an issue, partnering with industry to understand the most important materials and functions of essential goods will be key to agility in the future. As we have seen with the establishment of the Manufacturing USA network and the biomedical-specific institutes such as ARMI, NIIMBL, and BioMADE, working closely across sectors facilitates the sharing and implementation of best practices, standards, and guidance to create a nimbler infrastructure that can withstand and adjust to supply-chain constraints.
COVID-19 has left no industry or activity untouched, but the pandemic continues to negatively impact certain sectors more than others. Despite the importance of the research, development, and products coming out of the biomedical sector, policy decisions continue to have far-reaching implications on the sector’s ability to bounce back.
Though much has been done to create a “new normal,” there is still a way to go. It will take a thoughtful, considered response to ensure the biomedical sector can produce the results we all rely on.
Kevin D. Costa is the director of cardiovascular cell and tissue engineering at the Cardiovascular Research Institute of the Icahn School of Medicine at Mount Sinai in New York City. Costa was also the chair of the ASME Bioengineering Public Policy Task Force (BPPTF). Charles Y. Lee is an ASME Fellow and the director of the Center for Biomedical Engineering and Science at the University of North Carolina at Charlotte. Lee was the ASME Committee on Government Relations Liaison to the BPPTF.
The authors would like to acknowledge the other members of the task force for their contributions: Martin L. Tanaka, P.E., of Western Carolina University in Cullowhee, N.C.; Joe Budzinski, director of the Global Development Center at Johnson & Johnson; Roger Narayan of North Carolina State University in Raleigh; C. Ross Ethier, chair in bioengineering at the Georgia Institute of Technology and the Emory University School of Medicine; and Mike Hooven, CEO at Enable Injections.












