Vortex Ultrasound Tool Destroys Blood Clots from Within
Vortex Ultrasound Tool Destroys Blood Clots from Within


A research team across multiple institutions has developed a new tool and technique that uses vortex ultrasound to break down blood clots.
When it comes to eliminating blood clots of a certain size, current methods can only do so much. Existing treatment methods fail in as many as 20 percent to 40 percent of cerebral venous sinus thrombosis (CVST) cases, where clots form in the veins that drain blood away from the brain. But a multi-institutional innovation shows promise as a way to obliterate blood clots from within by using vortex ultrasound or sonothrombolysis.
To do this, researchers have developed a small aperture ultrasound transducer that fits within a catheter at 9 French gauge (3 millimeters) or less that can go inside a vein and emit vortex ultrasound waves that have a helical wavefront. explained Xiaoning Jiang, co-corresponding author of “A Model of High-Speed Endovascular Sonothrombolysis with Vortex Ultrasound-Induced Shear Stress to Treat Cerebral Venous Sinus Thrombosis,” published in Research.
“Thrombolysis, or using ultrasound to disrupt blood clots, has been in our interest for a while,” said Jiang, who is the Dean F. Duncan Professor of Mechanical and Aerospace Engineering at North Carolina State University. “We have been working on our existing grant over the last five years or so. We found that in using the small aperture and low frequency ultrasound, we were able to significantly improve thrombolysis efficacy and safety.”
You Might Also Enjoy: Stanching Blood Flow with Microneedles
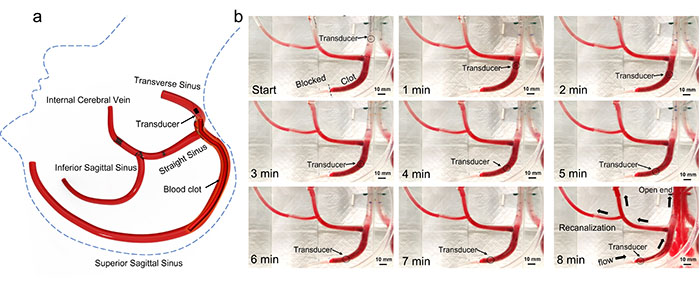
During the proof-of-concept in vitro testing, the team used cow blood inside of a 3D-printed model of the cerebral venous sinus. Their hypothesis was that vortex ultrasound would provide an advantage in terms of shear stress in combination with traditional cavitation or radiation force, which might help to enhance the thrombolysis efficacy, Jiang explained.
It worked. This technique has shown a tenfold increase in the lysis rate over existing drug-based or ultrasound-assisted drug penetration treatment methods during testing, Jiang added.
“For this particular application, we came up with single channel electronics, and were able to build four prototypes for this concept demonstration,” he said.
Current pharmaceutical interventions to dissolve CVST blood clots take at least 15 hours, and average around 29 hours, noted Chengzhi Shi, an assistant professor of mechanical engineering at Georgia Tech and co-corresponding author of the study. But, “during in vitro testing, we were able to dissolve an acute blood clot in well under half an hour,” he said.
A vortex ultrasound will induce a kind of sheer stress onto a blood clot, Shi added.
“So, what happens is, think about the structure of the blood clot is actually formed by these fibering structures that cluster all those red cells together and then form this clot that blocks the blood flow in the vessel,” Shi said. “Ultrasound is actually a pressure wave. If you draw the pressure low enough, you will form vapors or bubbles in the blood. And then because of the asymmetric distribution of the pressure, you will actually form some kind of micro jetting, so water from a kind of micro explosion of those bubbles, and then they will actuate those clots.”
Become a Member: How to Join ASME
But the problem is that the mass structure of fibers in the blood clot are resistant to normal stress, such as a traditional endovascular ultrasound, making that strategy less efficient and more time consuming to actuate a clot.
“This vortex ultrasound, because of the nature of the in-plane pressure gradient, will naturally induce the sheer stress. That's why we can break the block clot much faster, much more efficiently,” Shi said.
A frequency of 2 MHz has proven ideal thus far through testing, Shi added. Given the limited size of the aperture, the researchers can’t just choose any frequency at will. Instead, the device needs to target a frequency that will generate the vortex effect in a blood vessel more efficiently, Shi said. “To actually get to this frequency took a long time, a lot of simulations, and a lot of optimization.”
In another experiment, the researchers used the technology on a blood clot that was 7.5 cm long and 1 cm in diameter—dramatically larger than the typical 1-cm to 2-cm-long clots doctors might find in actual patients.
“There is no existing device that can actually treat this kind of severe case,” Shi said. “In our in vitro test, we also added pressure on the side of the clot to mimic low hypertension. And in that test, we show that we can recanalize, which means that we formed a channel for blood to restore the flow, within eight minutes for that case [with no damage to the simulated veins]. So that's a huge breakthrough if we can actually make it happen in the patient.”
Work began about two years ago, with more than a year going into developing and building the prototype vortex ultrasound transmitter and aperture, Jiang said. The effort has brought in researchers, students, clinicians, and others across multiple schools including North Carolina State University, Georgia Tech, UNC Chapel Hill, University of Michigan, and Pittsburgh University in this multi-institution, multi-disciplinary undertaking.
“Through that demonstration prototype, experiments, and a little more simulation, our hypothesis actually was confirmed: vortex ultrasound indeed can help improve thrombolysis rates. We also conducted some safety studies and haven't found any safety concerns associated with vortex ultrasound,” he added.
This isn’t the first such medical use of vortex ultrasound, but it is the first such application of its kind, the researchers noted.
Similar Reading: Engineers 3D Print Blood Vessel to Aid Vascular Disease Treatment
“Vortex ultrasound also creates a potential trap that can hold objects at the center of this wave,” Shi explained. A team at the University of Washington in collaboration with a team in Moscow, Russia, used vortex ultrasound as a trap to move kidney stones in a pig’s bladder. Another group of researchers in Taiwan tried to use this trap application to increase the efficiency of drug delivery into blood clots for lysis. “But we are the first ones who actually utilized the sheer stress induced by this vortex ultrasound to make it way more efficient,” Shi added.
Preliminary results have put the team in a better spot to target more funding, Jiang said. “When we have more resources, the next thing we want to do is optimize this small device or tornado catheter. And then we want to do an extensive animal study to verify the efficacy and the safety, and then hopefully by the end of that stage, this device will be ready for the clinical trial.”
Additional clinician involvement will be vital at that point.
“As engineers, we do the innovation, get started, concept demonstrated, but really to move from that, translate that kind of research into clinical practice, it takes time and resources,” Jiang said.
Louise Poirier is senior editor.
To do this, researchers have developed a small aperture ultrasound transducer that fits within a catheter at 9 French gauge (3 millimeters) or less that can go inside a vein and emit vortex ultrasound waves that have a helical wavefront. explained Xiaoning Jiang, co-corresponding author of “A Model of High-Speed Endovascular Sonothrombolysis with Vortex Ultrasound-Induced Shear Stress to Treat Cerebral Venous Sinus Thrombosis,” published in Research.
“Thrombolysis, or using ultrasound to disrupt blood clots, has been in our interest for a while,” said Jiang, who is the Dean F. Duncan Professor of Mechanical and Aerospace Engineering at North Carolina State University. “We have been working on our existing grant over the last five years or so. We found that in using the small aperture and low frequency ultrasound, we were able to significantly improve thrombolysis efficacy and safety.”
You Might Also Enjoy: Stanching Blood Flow with Microneedles
During the proof-of-concept in vitro testing, the team used cow blood inside of a 3D-printed model of the cerebral venous sinus. Their hypothesis was that vortex ultrasound would provide an advantage in terms of shear stress in combination with traditional cavitation or radiation force, which might help to enhance the thrombolysis efficacy, Jiang explained.
It worked. This technique has shown a tenfold increase in the lysis rate over existing drug-based or ultrasound-assisted drug penetration treatment methods during testing, Jiang added.
“For this particular application, we came up with single channel electronics, and were able to build four prototypes for this concept demonstration,” he said.
Current pharmaceutical interventions to dissolve CVST blood clots take at least 15 hours, and average around 29 hours, noted Chengzhi Shi, an assistant professor of mechanical engineering at Georgia Tech and co-corresponding author of the study. But, “during in vitro testing, we were able to dissolve an acute blood clot in well under half an hour,” he said.
A vortex ultrasound will induce a kind of sheer stress onto a blood clot, Shi added.
“So, what happens is, think about the structure of the blood clot is actually formed by these fibering structures that cluster all those red cells together and then form this clot that blocks the blood flow in the vessel,” Shi said. “Ultrasound is actually a pressure wave. If you draw the pressure low enough, you will form vapors or bubbles in the blood. And then because of the asymmetric distribution of the pressure, you will actually form some kind of micro jetting, so water from a kind of micro explosion of those bubbles, and then they will actuate those clots.”
Become a Member: How to Join ASME
But the problem is that the mass structure of fibers in the blood clot are resistant to normal stress, such as a traditional endovascular ultrasound, making that strategy less efficient and more time consuming to actuate a clot.
“This vortex ultrasound, because of the nature of the in-plane pressure gradient, will naturally induce the sheer stress. That's why we can break the block clot much faster, much more efficiently,” Shi said.
A frequency of 2 MHz has proven ideal thus far through testing, Shi added. Given the limited size of the aperture, the researchers can’t just choose any frequency at will. Instead, the device needs to target a frequency that will generate the vortex effect in a blood vessel more efficiently, Shi said. “To actually get to this frequency took a long time, a lot of simulations, and a lot of optimization.”
In another experiment, the researchers used the technology on a blood clot that was 7.5 cm long and 1 cm in diameter—dramatically larger than the typical 1-cm to 2-cm-long clots doctors might find in actual patients.
“There is no existing device that can actually treat this kind of severe case,” Shi said. “In our in vitro test, we also added pressure on the side of the clot to mimic low hypertension. And in that test, we show that we can recanalize, which means that we formed a channel for blood to restore the flow, within eight minutes for that case [with no damage to the simulated veins]. So that's a huge breakthrough if we can actually make it happen in the patient.”
Work began about two years ago, with more than a year going into developing and building the prototype vortex ultrasound transmitter and aperture, Jiang said. The effort has brought in researchers, students, clinicians, and others across multiple schools including North Carolina State University, Georgia Tech, UNC Chapel Hill, University of Michigan, and Pittsburgh University in this multi-institution, multi-disciplinary undertaking.
“Through that demonstration prototype, experiments, and a little more simulation, our hypothesis actually was confirmed: vortex ultrasound indeed can help improve thrombolysis rates. We also conducted some safety studies and haven't found any safety concerns associated with vortex ultrasound,” he added.
This isn’t the first such medical use of vortex ultrasound, but it is the first such application of its kind, the researchers noted.
Similar Reading: Engineers 3D Print Blood Vessel to Aid Vascular Disease Treatment
“Vortex ultrasound also creates a potential trap that can hold objects at the center of this wave,” Shi explained. A team at the University of Washington in collaboration with a team in Moscow, Russia, used vortex ultrasound as a trap to move kidney stones in a pig’s bladder. Another group of researchers in Taiwan tried to use this trap application to increase the efficiency of drug delivery into blood clots for lysis. “But we are the first ones who actually utilized the sheer stress induced by this vortex ultrasound to make it way more efficient,” Shi added.
Preliminary results have put the team in a better spot to target more funding, Jiang said. “When we have more resources, the next thing we want to do is optimize this small device or tornado catheter. And then we want to do an extensive animal study to verify the efficacy and the safety, and then hopefully by the end of that stage, this device will be ready for the clinical trial.”
Additional clinician involvement will be vital at that point.
“As engineers, we do the innovation, get started, concept demonstrated, but really to move from that, translate that kind of research into clinical practice, it takes time and resources,” Jiang said.
Louise Poirier is senior editor.